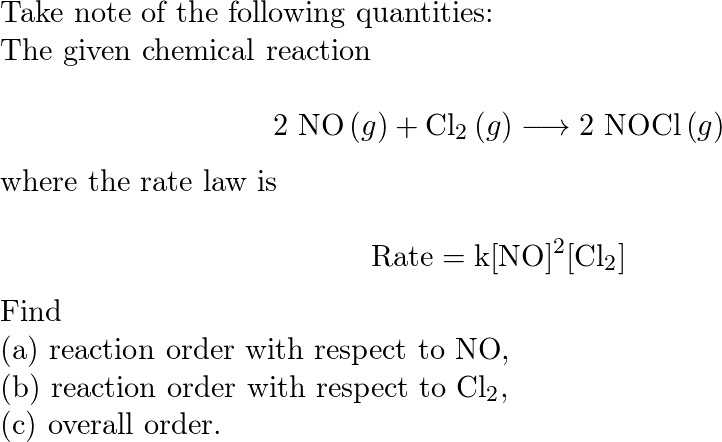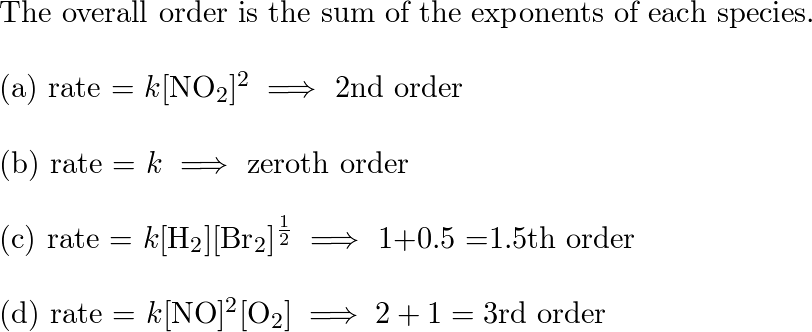Milwaukee Men's 2X-Large Black No Days Off Hooded Sweatshirt with Black Cuffed Knit Hat 311B-2X-503B - The Home Depot

Zenport J6-207 Aluminum handled hoe 55-inch overall length-no broken box orders (see notes o, 1 - Kroger

AMZ Supply Disposable Coveralls 71" Long Protective Orange PPE Protective Suit 2X-Large Pack of 25 - Walmart.com

Amazon.com: National Safety Apparel KIT2CV11NG2X ArcGuard CAT 2 Arc Flash Kit with FR Coverall (No Gloves), 12 Calorie, XX-Large, Navy : Tools & Home Improvement
![SOLVED: Classify each rate law based on whether the reaction is first-order; second-order; O third-order overall: First-order Second-order Third-order Answer Bank rate k[HCN] rate k[O] [NO] [NzE rate [BFs] [NH,] rate kINO]? SOLVED: Classify each rate law based on whether the reaction is first-order; second-order; O third-order overall: First-order Second-order Third-order Answer Bank rate k[HCN] rate k[O] [NO] [NzE rate [BFs] [NH,] rate kINO]?](https://cdn.numerade.com/ask_previews/7a671ad0-bb32-4650-a5e2-0dc410a20fb1_large.jpg)
SOLVED: Classify each rate law based on whether the reaction is first-order; second-order; O third-order overall: First-order Second-order Third-order Answer Bank rate k[HCN] rate k[O] [NO] [NzE rate [BFs] [NH,] rate kINO]?
![SOLVED: What is the overall reaction order for the reaction that has the rate law Rate = k[O2][NO]2? zero order first order second order third order SOLVED: What is the overall reaction order for the reaction that has the rate law Rate = k[O2][NO]2? zero order first order second order third order](https://cdn.numerade.com/ask_previews/bea16924-e043-4592-9ba1-7c64b3c6ae29_large.jpg)
SOLVED: What is the overall reaction order for the reaction that has the rate law Rate = k[O2][NO]2? zero order first order second order third order
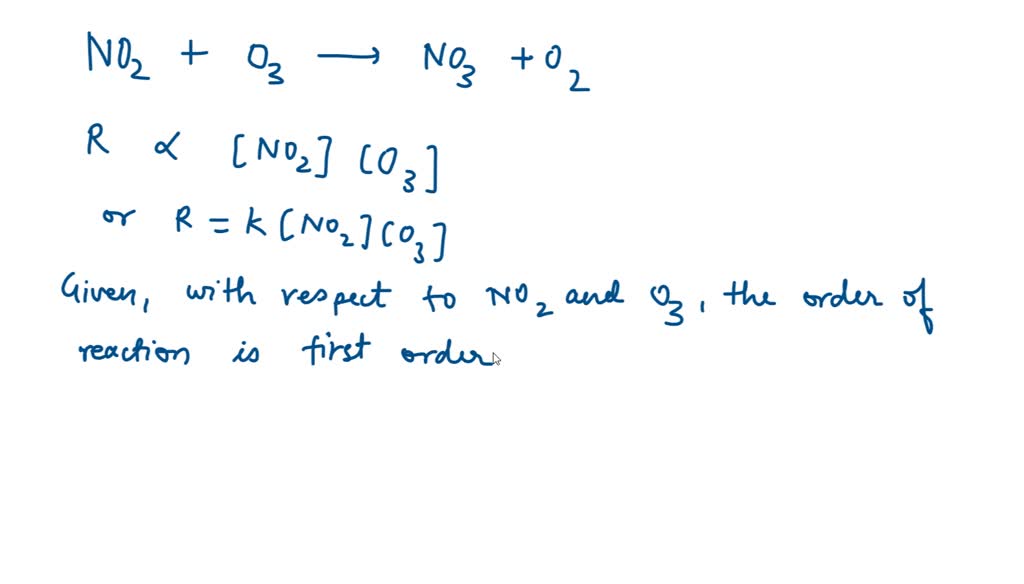
SOLVED: The reaction of NO2 with ozone O3 produces NO3 and O2 in a second order reaction overall. The rate law for the reaction if the reaction is first-order in each reactant

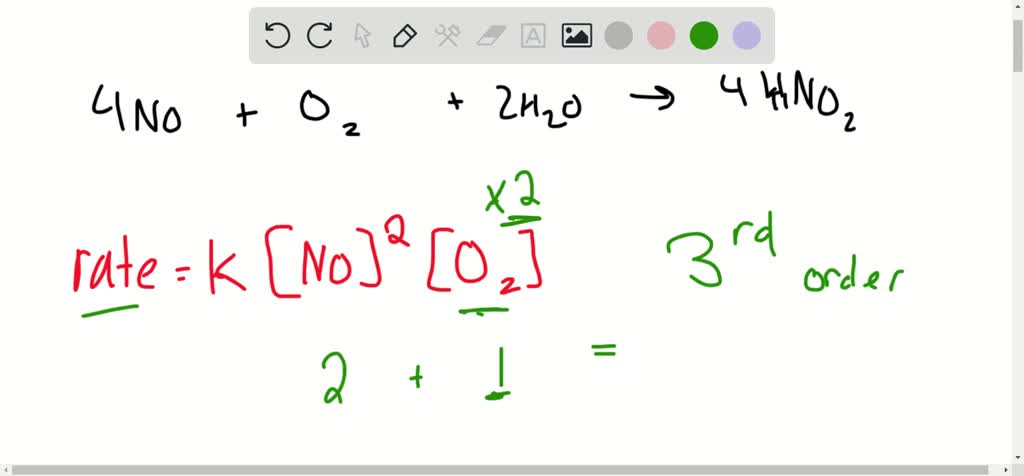
The rate of reaction , 2NO + Cl2 → 2NOCl is doubled when concentration of Cl2 is doubled and it becomes eight times when concentration of both NO and Cl2 are doubled .
![SOLVED: Question 3 (1 point) If m=-1 and n-2,then the overall order of reaction is: Question 4 (3 points) Experiment Initial Rate (M s-1) [NOz] (M) A 0.0050 0.10 B 0.0800 0.40 SOLVED: Question 3 (1 point) If m=-1 and n-2,then the overall order of reaction is: Question 4 (3 points) Experiment Initial Rate (M s-1) [NOz] (M) A 0.0050 0.10 B 0.0800 0.40](https://cdn.numerade.com/ask_previews/80ab0b03-b365-4adf-aecd-bfc5fcfa3766_large.jpg)
SOLVED: Question 3 (1 point) If m=-1 and n-2,then the overall order of reaction is: Question 4 (3 points) Experiment Initial Rate (M s-1) [NOz] (M) A 0.0050 0.10 B 0.0800 0.40

3M - All-Purpose & Utility Hooks; Overall Length (Inch): 1-1/4; Material: Plastic; Projection: 0.50 in; Projection: 0.50; Width (Inch): 0.82 in; Minimum Order Quantity: Plastic; Type: Clips Kit; Material: Plastic; Description: Warm
![SOLVED: What is the overall order of the reaction below CO (g) + NO2 (g) -> CO2 (g) + NO (g) if it proceeds via the following rate expression? Rate = k[CO][NO2] SOLVED: What is the overall order of the reaction below CO (g) + NO2 (g) -> CO2 (g) + NO (g) if it proceeds via the following rate expression? Rate = k[CO][NO2]](https://cdn.numerade.com/ask_previews/8a209c4b-4b10-42a8-a36b-2cfd44a34aac_large.jpg)
SOLVED: What is the overall order of the reaction below CO (g) + NO2 (g) -> CO2 (g) + NO (g) if it proceeds via the following rate expression? Rate = k[CO][NO2]